Table Of Content
Otherwise, the amount of preliminary data necessary will depend on the trial phase. For instance, in the early phases of a tDCS trial, the investigators tested the device in 8 PLP subjects with one session only, looking for immediate effects and testing different parameters (10). This, combined with further data on brain stimulation in pain, led the same group to then design a large pivotal trial using optimized parameters (25). We will also cover the gold standard for analysis of clinical trials, which is including all the participants in the analysis regardless of their actual treatment.
Professional development
We know this subject lived ≥5 years and this can be used to evaluate and estimate survival up to 5 years. After 5 years the subject cannot contribute data for estimating or quantifying survival and they are termed censored. Different statistical analyses are needed for time-to-event versus quantitative data to account for censoring. Clinicians and other decision makers in healthcare use results from clinical trials to inform practice.
Publication types
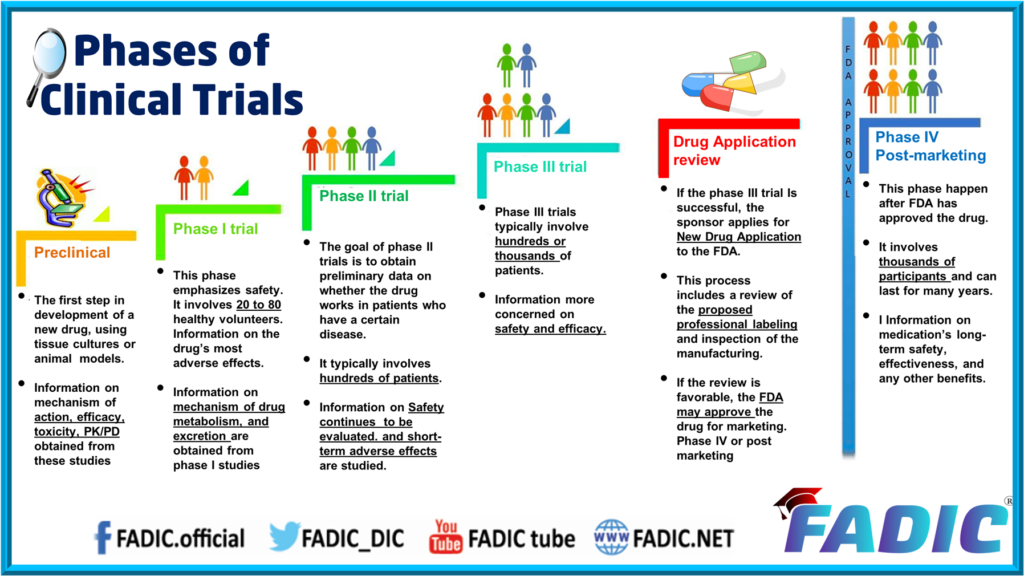
Phase II trials aim to further understand the safety and efficacy of an intervention to help decide whether or not to proceed to a phase III trial. Given the role of Phase II trials in determining the go/no-go decision to proceed for further testing in large confirmatory Phase III trials, it is crucial to select an appropriate endpoint, particularly in Phase II trials. Phase II endpoints should ideally be a strong surrogate for the Phase III endpoints (Yin et al., 2018). The delivery of an intervention whether drug, a dietary change, a lifestyle change, or a psychological therapy session counts as an intervention and hence must be dealt as a clinical trial [Figure 1].

Code, Data and Media Associated with this Article
Study analysis is conducted using only data from the withdrawal phase and outcome is usually relapse of symptoms. The randomized withdrawal design aims to evaluate the optimal duration of a treatment in patients who respond to the treatment. The advantage is reduction in the time on placebo since only responders are randomized to placebo thereby giving an ethical advantage.
Designing studies with a strong foundation I: preliminary clinical data
Researchers could then consider using a sham injection that induces a similar reaction. Variation can also be reduced with standardization of the manner in which study participants are treated and evaluated via training. For example, in studies that involve imaging, it is very important to have an imaging protocol that standardizes the manner in which images are collected to reduce added variation due to inconsistent patient positioning. Training modules can be developed to instruct site personnel on the appropriate administration of evaluations. We have discussed various clinical research study designs in this comprehensive review.
Having “double-blind” in the title of a trial does not imply that blinding was successful. Reviews of blinded trials suggest that many trials experience issues that jeopardize the blind. For example in a study assessing zinc for the treatment of the common cold(Prasad et al 2000) the blinding failed because the taste and aftertaste of zinc was distinctive. For example, OHARA and the ACTG are developing a study to evaluate the use of gentian violet (GV) for the treatment of oral candidiasis. GV has staining potential which could jeopardize the blind when the assessors conduct oral examinations after treatment.
Clinical Trial Design and Colonoscopy Prep for Patients With IBD - Gastroenterology & Endoscopy News
Clinical Trial Design and Colonoscopy Prep for Patients With IBD.
Posted: Mon, 11 Dec 2023 08:00:00 GMT [source]
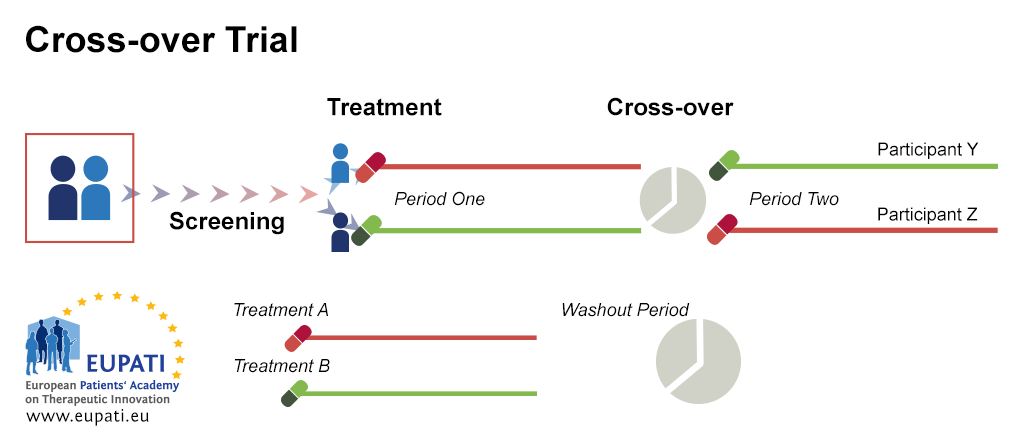
(a) The disease must be chronic, stable, and incurable and characteristics must not vary for the duration of the two study periods and the interim wash out period and (b) the effect of each drug must not be irreversible. Bioequivalence and biosimilar equivalence studies usually utilize a cross over design. The duration of follow-up for the patient is longer than for a parallel design, and there is a risk that a significant number of patients do not complete the study and drop out leading to compromised study power. Treatment effects are efficiently isolated by controlling for bias and confounding and by minimizing variation.
6 Selection of a control group
Once the rationale for a trial has been established, selection of the outcome(s) of interest is essential. Examples of health outcomes include quality of life, symptoms, adverse events, and patient-reported outcomes. Treatment outcomes include assessing safety or efficacy of the intervention; examples include tumor shrinkage, hematologic outcomes, intermediate or surrogate outcomes, time to event outcomes (e.g. overall survival or progression-free survival), and surgical outcomes. It is common to have one or two primary outcomes, and one or two secondary outcomes.
Related Subjects
However, in a case series, the cases are not compared to subjects without the manifestations and therefore it cannot determine which factors in the description are unique to the new disease entity. In summary, a benefit-risk assessment is always necessary when designing a trial, and the benefits and risks to research subjects vs. those for the target population are crucial to identify. Using a Bayesian analytic framework, between 40 and 125 patients are enrolled for each therapeutic arm, with pre-specified criteria for graduation (that is, declaring a therapy to be likely efficacious) or futility.
Well-designed negative trials can generate useful data, and have been published in high-impact journals (26). Trial failure means that results are not valid and so we are unable to either reject or not reject the null hypothesis. Phase II trials are also important to collect additional safety data, determining drug dosing ranges, routes and timing for phase III trials, as well as common short-term adverse events. There are numerous phase II trials evaluating the efficacy of pharmacological agents in PLP, for example, gabapentin (17), ketamine (18), memantine (19), and calcitonin combined with ketamine (20).
The advantage of this design is its flexibility such that it can be continued until a definitive conclusion can be reached for the particular subject being studied. The utility also rests in analyzing treatments that elicit heterogenous responses in different subjects. Data from many N-of 1 subjects can be even combined to derive population effect sizes by meta-analysis or Bayesian methods. Common clinical trial designs include single-arm trials, placebo-controlled trials, crossover trials, factorial trials, noninferiority trials, and designs for validating a diagnostic device. The choice of the structural design depends on the specific research questions of interest, characteristics of the disease and therapy, the endpoints, the availability of a control group, and on the availability of funding. Structural designs are discussed in an accompanying article in this special issue.
Randomization can be balanced where both groups are of equal size or unbalanced where groups are of unequal size. Finally, when confounding factors may be of concern, stratification may be considered as an additional design component. Although randomization aims to reduce confounding by making treatment groups as similar as possible except for the treatment assigned, it is nevertheless possible for the groups to differ with respect to some important factors. Examples of such factors include gender and age, and other factors specific to the study context. To avoid this possibility, identify these potential confounding factors and include stratification as part of the randomization process.
Finally, we will review the essential ethical consideration involved in conducting experiments on people. When blinding is implemented in a clinical trial, a plan for assessing the effectiveness of the blinding may be arranged. This usually requires two blinding questionnaires, one completed by the trial participant and the other completed by the local investigator or person that conducts the evaluation of the trial participant.
A randomized clinical trial represents a controlled experiment in which the investigator applies an intervention to a group of patients and subsequently observes its effect on 1 or more outcomes over time. In this article, we outline the key elements that need to be considered during the design phase of a surgical clinical trial (Box). An active control is an active intervention that has often shown effectiveness to treat the disease under study. Often an active control is selected because it is the standard of care (SOC) treatment for the disease under study.
CIT can broadcast your seminar, conference or meeting live to a world-wide audience over the Internet as a real-time streaming video. The event can be recorded and made available for viewers to watch at their convenience as an on-demand video or a downloadable file. Some of the considerations here are shared under the more general topic of design of experiments but there can be others, in particular related to patient confidentiality and ethics. Some of the authors have been involved in a number of the trials referenced in this paper.
Objective endpoints are generally preferred to subjective endpoints since they are less subject to bias. The most common is hypothesis testing where researchers construct a null hypothesis (often “no effect” or “no difference”) that is assumed to be true and evidence is sought to disprove it. An alternative hypothesis (the statement that is desired to be claimed) is also constructed (often the presence of an effect or difference between groups).
No comments:
Post a Comment